|
11/25/2023 0 Comments Change in entropyQ is positive for the cold water, because heat was added, and negative for the hot water. The heat transferred from the hot water to the cold water is therefore:Ĭalculate the change in entropy for the hot and cold water using the equation: The process is irreversible - any process involving a transfer of heat from a higher-temperature region to a lower-temperature region is irreversible.Īssuming no heat is exchanged with the surroundings or the environment, what is the change in entropy in the mixing process?įirst, determine how much heat is involved. Is this process reversible or irreversible? 
The colder water is then poured into the warmer water, and the system is allowed to come to equilibrium. In one the water temperature is 17☌, while in the other it is 37☌. You have two styrofoam containers of water. Time moves in the direction of increasing entropy. This is why the glass of spilled milk never spontaneously transforms itself back into an upright full glass of milk - that would decrease the entropy.Įntropy is often called time's arrow. The entropy of a closed system is constant for reversible processes and increases for irreversible processes. The Second Law of Thermodynamics states that: In these there is no change in entropy in a closed system. The entropy postulate connects the concept of entropy with such processes:Įntropy Postulate: If an irreversible process occurs in a closed system, the entropy S of the system always increases. This is an example of an irreversible process. Even though Newton's Laws and The Laws of Conservation of Energy and Conservation of Momentum would be obeyed when you played the film backwards, the probability that all the milk and the glass would spontaneously come together to form a full glass of milk is incredibly small. If you videotaped the spill and then played the film backwards, it would be obvious to you that the film was running backwards. If you spill a glass of milk, what the glass and the milk droplets do is governed by the laws of physics. ΔS = nR ln(V f / V i) + nC V ln(T f / T i) If the heat transfer takes place over a range of temperatures then, as long as ΔT is small compared to the absolute temperature T, the change in entropy is approximately:įor an ideal gas, it can be shown that the change in entropy is given by: If the heat transfer takes place at a single temperature, the change in entropy is simply: The change in entropy is the heat added divided by the temperature at which the transfer took place. On the other, a change in entropy is easy to determine.Įntropy changes whenever there is a transfer of heat. Unlike P, V, and T, which are quite easy to measure, the entropy of a system is difficult to calculate.  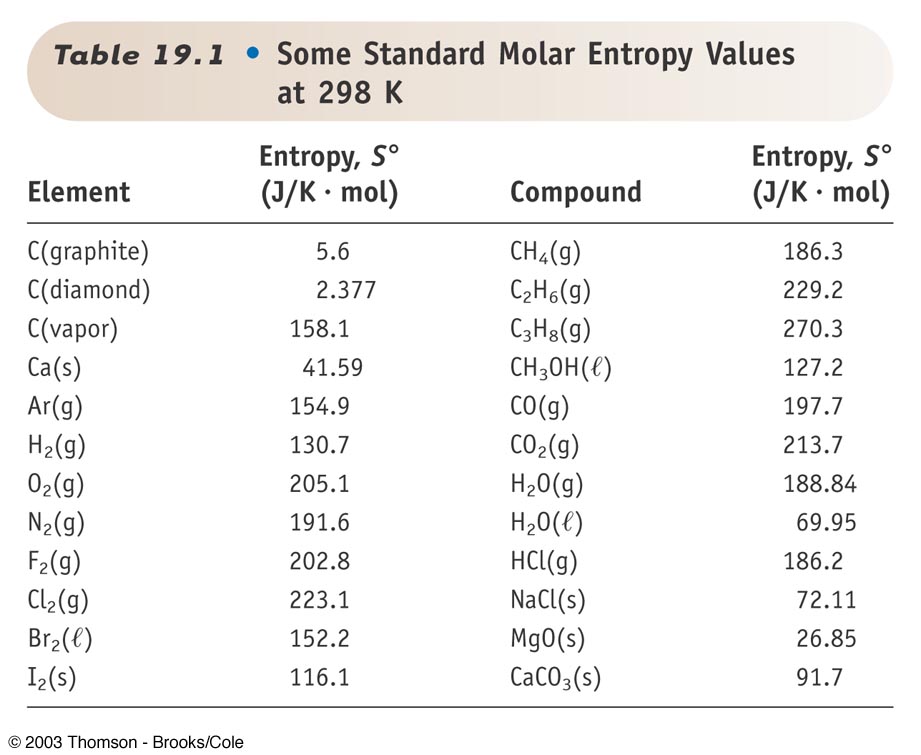
This is the basic way of evaluating S for constant-temperature processes such as phase changes, or the isothermal expansion of a gas. The symbol for entropy is S, and the units are J/K.Ī container of ideal gas has an entropy value, just as it has a pressure, a volume, and a temperature. For a process that reversibly exchanges a quantity of heat qrev with the surroundings, the entropy change is defined as. Here we further explore the nature of this state function and define it mathematically.Entropy is in some sense a measure of disorder. In Chapter 13, we introduced the concept of entropy in relation to solution formation. To help explain why these phenomena proceed spontaneously in only one direction requires an additional state function called entropy (S), a thermodynamic property of all substances that is proportional to their degree of "disorder". that is, the entropy change of a thermodynamic process is equal to the generated entropy in the process boundary plus the net entropy transferred to the process. Moreover, the molecules of a gas remain evenly distributed throughout the entire volume of a glass bulb and never spontaneously assemble in only one portion of the available volume. For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
